According to research studies, transcranial infrared laser stimulation, as well as other types of transcranial lasers, utilized on frontal cortex functions can improve sustained attention and working memory, among other brain functions. Transcranial laser stimulation with low-power density (mW/cm2) and high-energy density (J/cm2) monochromatic light in the near-infrared wavelengths regulates and maintains brain functions and may promote neurotherapeutic effects in a non-destructive and non-thermal manner. Researchers determined through the first controlled research study that transcranial laser stimulation improves human cognitive and emotional brain functions. Â
In the field of low-level light/laser therapy or LLLT, developing a model to demonstrate how luminous energy from red-to-near-infrared wavelengths improves bioenergetics has been in development for the last 40 years. Previous LLLT research studies have demonstrated historical a variety of developments, principles, and applications on the subject matter. The purpose of the following article is to demonstrate an update on LLLT’s neurochemical mechanisms supporting transcranial laser stimulation for cognitive-enhancing functions. We will describe the effect of LLLT on brain bioenergetics, briefly discussing its bioavailability and dose-response, and its effects on cognitive brain function. Although our focus is on prefrontal-related cognitive functions, LLLT should be able to improve other brain functions. By way of instance, stimulating different brain regions affect different functions associated with sensory and motor systems. Â
Transcranial Lasers on Brain Bioenergetics
Near-infrared lasers and light-emitting diodes, or LEDs, affect brain function according to bioenergetics, a mechanism which is fundamentally different than other brain stimulation methods and techniques, such as electric and magnetic stimulation. LLLT has been demonstrated to regulate and maintain the function of neurons in cell cultures, and brain function in animals as well as cognitive and emotional functions in patients and health issues. Photoneuromodulation is associated with the absorption of photons by certain molecules in neurons which activate bioenergetic signaling pathways after being exposed to red-to-near-infrared light. The 600mm to 1150 nm wavelengths provide improved tissue penetration by photons because light is scattered at lower wavelengths and absorbed by water at higher wavelengths. Over 25 years ago, it was demonstrated that molecules which absorb LLLT wavelengths are part of the mitochondrial respiratory enzyme cytochrome oxidase in different oxidation states. Moreover, for red-to-near-infrared light, the main molecular photoacceptor of photon energy is cytochrome oxidase, also known as cytochrome c oxidase or cytochrome a-a3. Â
Furthermore, photon energy absorption by cytochrome oxidase is the main neurochemical mechanism of action of LLLT in neurons. The more the enzymatic activity of cytochrome oxidase increases, the more metabolic energy which is developed through mitochondrial oxidative phosphorylation. LLLT provides the brain with metabolic energy in an analogous manner to the conversion of nutrients into metabolic energy with the utilization of light instead of nutrients developing the source for ATP-based metabolic energy. If an effective near-infrared light energy dose is provided, it stimulates brain ATP production and blood flow, ultimately fueling ATP-dependent membrane ion pumps, promoting greater membrane stability and resistance to depolarization, which has been demonstrated to transiently reduce neuronal excitability. Electromagnetic stimulation also directly affects the electrical excitability of neurons, as demonstrated in research studies. Â
A long-lasting effect is provided by LLLT’s up-regulating the amount of cytochrome oxidase, which improved neuronal capacity for metabolic energy production which can be utilized to improve cognitive brain functions. In mice and rats, memory has been demonstrated to improve by LLLT and by methylene blue, a drug, which at low doses, provides electrons to cytochrome oxidase. Near-infrared light stimulates mitochondrial respiration by providing photons to cytochrome oxidase because cytochrome oxidase mainly accepts photons from red-to-near-infrared light in neurons. By stimulating cytochrome oxidase activity, transcranial LLLT promotes post-stimulation up-regulation of the amount of cytochrome oxidase in brain mitochondria. LLLT may also improve the conversion of luminous energy into metabolic energy during light exposure as well as the up-regulation of the mitochondrial enzymatic machinery to develop more energy after light exposure. Â
Bioavailability and Hormetic Dose-Response by Transcranial Lasers
The most numerous metalloprotein found in nerve tissue is cytochrome oxidase and its absorption wavelengths are often associated with its enzymatic activity and ATP production. Increased LLLT bioavailability to the brain in vivo has been demonstrated in a variety of research studies by exposing brain cytochrome oxidase activity transcranially, resulting in improved extinction memory retention in healthy rats and improved visual discrimination in rats with impaired retinal mitochondrial function. Other LLLT research studies utilized a variety of wavelengths (633–1064 nm), daily doses (1–60 J/cm2), fractionation sessions (1–6), and power densities (2–250 mW/cm2) which ultimately characterized effective LLLT parameters for both rats and humans. Â
By way of instance, researchers evaluated in rats the effects of different LLLT doses in vivo on brain cytochrome oxidase activity, at either 10.9, 21.6, 32.9 J/cm2, or no LLLT. Treatments were utilized for 20, 40, and 60 min through four 660-nm LED arrays with a power density of 9 mW/cm2. One day after the LLLT session, the brains of the rats were extracted, frozen, sectioned, and processed for cytochrome oxidase histochemistry. A 10.9 J/cm2 dose increased cytochrome oxidase activity by 13.6 percent. A 21.6 J/cm2 dose developed a 10.3 percent increase. A non-significant cytochrome oxidase increase of 3 percent was found after the highest 32.9 J/cm2 dose. Responses of brain cytochrome oxidase to LLLT in vivo were characterized by hormesis, with a low dose being stimulatory while higher doses were less effective. Â
The first demonstration that LLLT increased oxygen consumption in the rat prefrontal cortex in vivo was demonstrated by another research study. Oxygen concentration in the cortex of rats was measured utilizing fluorescence-quenching during LLLT at 9 mW/cm2 and 660 nm. LLLT promoted a dose-dependent increase in oxygen consumption of 5 percent after 1 J/cm2 and 16 percent after 5 J/cm2. Because oxygen is utilized to develop water within mitochondria in a response developed by cytochrome oxidase, more cytochrome oxidase activity should promote more oxygen consumption. Â
LLLT can also offer several benefits over other types of stimulation because LLLT non-invasively targets cytochrome oxidase, a fundamental enzyme utilized for energy production, with promoted expression associated with energy increase. LLLT is mechanistically specific and non-invasive while transcranial magnetic stimulation may be non-specific, prolonged forehead electrical stimulation may increase muscle spasms and deep brain or vagus nerve stimulations are invasive. Â
Transcranial Lasers on Cognitive and Emotional Functions
LLLT through commercial low-power sources, such as FDA-cleared laser diodes and LEDs, is a highly promising, affordable, non-pharmacological alternative treatment option for improving cognitive brain function. LLLT offers safe doses of light energy which regulate and maintain neuronal functions, however, these are low enough to not damage the brain. In 2002, the FDA approved LLLT for pain relief in cases of head and neck pain, arthritis and carpal tunnel syndrome. LLLT has been utilized non-invasively in humans after ischemic stroke to improve neurological functions. It also improved recovery and ultimately reduced fatigue after exercise. One LLLT stimulation session to the forehead, as demonstrated in another research study, developed a considerable antidepressant effect in patients with depression. No adverse side effects were found either immediately nor at 2 or 4 weeks after LLLT. Therefore, LLLT treatments have been demonstrated to be safe and effective in humans. Although LLLT has been determined to be safe and it received FDA approval to be utilized for pain treatment, transcranial lasers for the augmentation of cognitive brain function should be limited for further research studies until further outcome measures support this application for clinical utilization. Â
Transcranial laser stimulation to the forehead was utilized in a placebo-controlled, randomized research study, to demonstrate the effects of cognitive tasks associated with the prefrontal cortex, including a psychomotor vigilance task, or PVT, and a delayed match-to-sample, or DMS, memory task. The PVT evaluates sustained attention, with patients remaining vigilant during delay intervals, and pushing a button when visual stimulation appeared on a monitor. The laser stimulation targeted prefrontal regions which are believed to be utilized in the sustained attentional processes of the PVT. The DMS task supports the prefrontal cortex as part of a network of frontal and parietal brain regions. Â
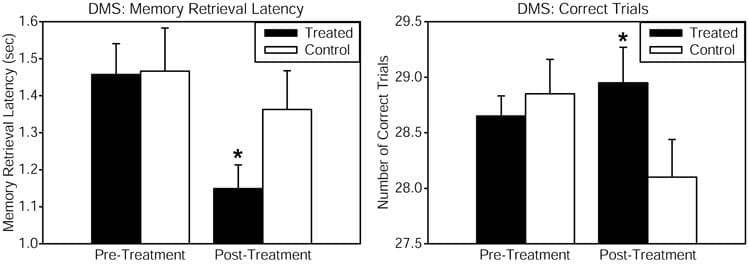
Healthy patients received consistent wave near-infrared light intersecting cytochrome oxidase’s absorption spectrum, targetted to the forehead utilizing a 1064 nm low-power laser diode, also known as “cold laserâ€, which increases tissue penetration due to its long wavelength and has been utilized in humans for other health issues. The power density or irradiance, 250 mW/cm2, and the cumulative energy density or fluence, 60 J/cm2, were identical which demonstrated the benefits of psychological effects in another research study. This laser exposure develops negligible heat and no physical damage at the low power level utilized. This laser apparatus is utilized safely in a clinical setting by the supplier of the laser. Reaction time in the PVT was improved by the laser treatment, as demonstrated by a considerable pre-post reaction time effect associated with the placebo group. The DMS memory task also demonstrated considerable improvements in measures of memory retrieval latency and number of correct trials, when comparing the LLLT-treated with the placebo group as demonstrated in Figure 1. Self-reported positive and negative affective or emotional states were also measured utilizing the PANAS-X questionnaire before and 2 weeks after laser treatment. As compared to the placebo, treated patients demonstrated considerably improved affective states. We suggest that this type of transcranial laser stimulation may serve as a non-invasive and efficacious method and technique to augment cognitive brain functions associated with attention, memory, and emotional functions. Â
LLLT’s bioenergetics mechanisms associated with cognitive augmentation may also be associated in its neuroprotective effects. LLLT’s stimulation of mitochondrial respiration should improve cellular function due to increased metabolic energy and cellular survival after injury due to the antioxidant effects of increases in cytochrome oxidase and superoxide dismutase. Â
Laser transmittance of the 1064-nm wavelength at the forehead LLLT site was estimated in a post-mortem human specimen, which demonstrated that approximately 2 percent of the light passed through the frontal bone. This yielded an absorption coefficient of a = 0.24, similar to the demonstrated a = 0.22 transmittance through cranial bone for this wavelength. Furthermore, it was estimated that about 1.2 J/cm2 of the 60 J/cm2 LLLT dose applied reached the surface of the prefrontal cortex. This value is similar to 1 J/cm2, the peak effective LLLT dose in neuron cultures for increasing cytochrome oxidase activity. Â
Transcranial absorption of photon energy by cytochrome oxidase, the terminal enzyme in mitochondrial respiration, is associated as the bioenergetic mechanism of action of LLLT in the brain. Transcranial LLLT up-regulates cortical cytochrome oxidase and improves oxidative phosphorylation. LLLT improves prefrontal cortex-related cognitive functions, such as sustained attention, extinction memory, working memory, and affective state. Transcranial infrared stimulation may be utilized effectively to support neuronal mitochondrial respiration as a new non-invasive, cognition-improving intervention in animals and humans. This fascinating new treatment approach should also be able to affect other brain functions associated with the neuroanatomical site stimulated and the stimulation parameters utilized. Â

Low-level laser therapy, or LLLT, and other types of transcranial lasers are non-invasive, low-powered lasers which are now being utilized in specific cortical regions of the brain to improve physiological responses and cognitive function. Many research studies have demonstrated that transcranial lasers can ultimately improve attention, memory, and reactions, where many other research studies have also demonstrated that these can also help improve depression and possibly even Alzheimer’s disease. Although further research studies are still required, the outcome measures are promising. – Dr. Alex Jimenez D.C., C.C.S.T. Insight
Diet and Exercise for Neurological Disease
According to research studies, transcranial infrared laser stimulation, as well as other types of transcranial lasers, utilized on frontal cortex functions can improve sustained attention and working memory, among other brain functions. Transcranial laser stimulation regulates and maintains brain functions and may promote neurotherapeutic effects in a non-destructive and non-thermal manner. Researchers determined through the first controlled research study that transcranial laser stimulation improves human cognitive and emotional brain functions. The scope of our information is limited to chiropractic, musculoskeletal and nervous health issues as well as functional medicine articles, topics, and discussions. To further discuss the subject matter above, please feel free to ask Dr. Alex Jimenez or contact us at 915-850-0900 . Â
Curated by Dr. Alex Jimenez Â
Additional Topic Discussion: Chronic Pain
Sudden pain is a natural response of the nervous system which helps to demonstrate possible injury. By way of instance, pain signals travel from an injured region through the nerves and spinal cord to the brain. Pain is generally less severe as the injury heals, however, chronic pain is different than the average type of pain. With chronic pain, the human body will continue sending pain signals to the brain, regardless if the injury has healed. Chronic pain can last for several weeks to even several years. Chronic pain can tremendously affect a patient’s mobility and it can reduce flexibility, strength, and endurance.
Neural Zoomer Plus for Neurological Disease
Dr. Alex Jimenez utilizes a series of tests to help evaluate neurological diseases. The Neural ZoomerTM Plus is an array of neurological autoantibodies which offers specific antibody-to-antigen recognition. The Vibrant Neural ZoomerTM Plus is designed to assess an individual’s reactivity to 48 neurological antigens with connections to a variety of neurologically related diseases. The Vibrant Neural ZoomerTM Plus aims to reduce neurological conditions by empowering patients and physicians with a vital resource for early risk detection and an enhanced focus on personalized primary prevention. Â
Formulas for Methylation Support
XYMOGEN’s Exclusive Professional Formulas are available through select licensed health care professionals. The internet sale and discounting of XYMOGEN formulas are strictly prohibited.
Proudly, Dr. Alexander Jimenez makes XYMOGEN formulas available only to patients under our care.
Please call our office in order for us to assign a doctor consultation for immediate access.
If you are a patient of Injury Medical & Chiropractic Clinic, you may inquire about XYMOGEN by calling 915-850-0900.
For your convenience and review of the XYMOGEN products please review the following link.*XYMOGEN-Catalog-Download Â
* All of the above XYMOGEN policies remain strictly in force.
Â
Post Disclaimers
General Disclaimer, Licenses and Board Certifications *
Professional Scope of Practice *
The information herein on "Transcranial Lasers for Cognitive Brain Function" is not intended to replace a one-on-one relationship with a qualified health care professional or licensed physician and is not medical advice. We encourage you to make healthcare decisions based on your research and partnership with a qualified healthcare professional.
Blog Information & Scope Discussions
Welcome to El Paso's Premier Wellness and Injury Care Clinic & Wellness Blog, where Dr. Alex Jimenez, DC, FNP-C, a Multi-State board-certified Family Practice Nurse Practitioner (FNP-BC) and Chiropractor (DC), presents insights on how our multidisciplinary team is dedicated to holistic healing and personalized care. Our practice aligns with evidence-based treatment protocols inspired by integrative medicine principles, similar to those on this site and on our family practice-based chiromed.com site, focusing on naturally restoring health for patients of all ages.
Our areas of multidisciplinary practice include Wellness & Nutrition, Chronic Pain, Personal Injury, Auto Accident Care, Work Injuries, Back Injury, Low Back Pain, Neck Pain, Migraine Headaches, Sports Injuries, Severe Sciatica, Scoliosis, Complex Herniated Discs, Fibromyalgia, Chronic Pain, Complex Injuries, Stress Management, Functional Medicine Treatments, and in-scope care protocols.
Our information scope is multidisciplinary, focusing on musculoskeletal and physical medicine, wellness, contributing etiological viscerosomatic disturbances within clinical presentations, associated somato-visceral reflex clinical dynamics, subluxation complexes, sensitive health issues, and functional medicine articles, topics, and discussions.
We provide and present clinical collaboration with specialists from various disciplines. Each specialist is governed by their professional scope of practice and their jurisdiction of licensure. We use functional health & wellness protocols to treat and support care for musculoskeletal injuries or disorders.
Our videos, posts, topics, and insights address clinical matters and issues that are directly or indirectly related to our clinical scope of practice.
Our office has made a reasonable effort to provide supportive citations and has identified relevant research studies that support our posts. We provide copies of supporting research studies upon request to regulatory boards and the public.
We understand that we cover matters that require an additional explanation of how they may assist in a particular care plan or treatment protocol; therefore, to discuss the subject matter above further, please feel free to ask Dr. Alex Jimenez, DC, APRN, FNP-BC, or contact us at 915-850-0900.
We are here to help you and your family.
Blessings
Dr. Alex Jimenez DC, MSACP, APRN, FNP-BC*, CCST, IFMCP, CFMP, ATN
email: [email protected]
Multidisciplinary Licensing & Board Certifications:
Licensed as a Doctor of Chiropractic (DC) in Texas & New Mexico*
Texas DC License #: TX5807, Verified: TX5807
New Mexico DC License #: NM-DC2182, Verified: NM-DC2182
Multi-State Advanced Practice Registered Nurse (APRN*) in Texas & Multi-States
Multi-state Compact APRN License by Endorsement (42 States)
Texas APRN License #: 1191402, Verified: 1191402 *
Florida APRN License #: 11043890, Verified: APRN11043890 *
Colorado License #: C-APN.0105610-C-NP, Verified: C-APN.0105610-C-NP
New York License #: N25929, Verified N25929
License Verification Link: Nursys License Verifier
* Prescriptive Authority Authorized
ANCC FNP-BC: Board Certified Nurse Practitioner*
Compact Status: Multi-State License: Authorized to Practice in 40 States*
Graduate with Honors: ICHS: MSN-FNP (Family Nurse Practitioner Program)
Degree Granted. Master's in Family Practice MSN Diploma (Cum Laude)
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
My Digital Business Card
Licenses and Board Certifications:
DC: Doctor of Chiropractic
APRNP: Advanced Practice Registered Nurse
FNP-BC: Family Practice Specialization (Multi-State Board Certified)
RN: Registered Nurse (Multi-State Compact License)
CFMP: Certified Functional Medicine Provider
MSN-FNP: Master of Science in Family Practice Medicine
MSACP: Master of Science in Advanced Clinical Practice
IFMCP: Institute of Functional Medicine
CCST: Certified Chiropractic Spinal Trauma
ATN: Advanced Translational Neutrogenomics
Memberships & Associations:
TCA: Texas Chiropractic Association: Member ID: 104311
AANP: American Association of Nurse Practitioners: Member ID: 2198960
ANA: American Nurse Association: Member ID: 06458222 (District TX01)
TNA: Texas Nurse Association: Member ID: 06458222
NPI: 1205907805
| Primary Taxonomy | Selected Taxonomy | State | License Number |
|---|---|---|---|
| No | 111N00000X - Chiropractor | NM | DC2182 |
| Yes | 111N00000X - Chiropractor | TX | DC5807 |
| Yes | 363LF0000X - Nurse Practitioner - Family | TX | 1191402 |
| Yes | 363LF0000X - Nurse Practitioner - Family | FL | 11043890 |
| Yes | 363LF0000X - Nurse Practitioner - Family | CO | C-APN.0105610-C-NP |
| Yes | 363LF0000X - Nurse Practitioner - Family | NY | N25929 |
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
My Digital Business Card











Comments are closed.